Precursor Synthesis and Characterisation
Synthesis of Precursors for Materials Synthesis
Our research is based on a methodology involving the conversion of molecular species to inorganic nanostructures and films through appropriate processes. Therefore, we synthesise these reactive molecules, such as metal alkoxides and metal amides, by Schlenk techniques and characterise them by several appropriate analysis methods (NMR, MS, IR, XRD…). Moreover, gas phase approaches require an appropriate partial pressure of the precursors. Hence, the degree of oligomerisation and intermolecular interactions for suitable precursors should be low to ensure appropriate volatility. Solution-based processes do not suffer from such restrictions and therefore the number of suitable precursor species is higher. Therefore, a larger number of precursor species can be used due to the simpler requirements such as solubility and decomposition characteristics. All metalorganic compounds used in the nanomaterial synthesis are prepared in our laboratory.
|
|
|
Precursors for Focused Electron-Induced Deposition
|
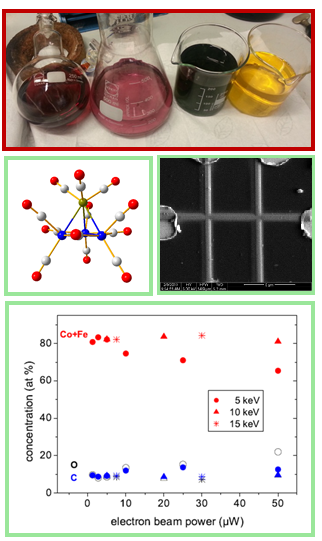
A new and highly interesting topic of our research is devoted to investigating molecules which can be used in focused electron beam induced deposition (FEBID) processes. This research on identification and synthesis of new proecursors is highly relevant, because most electron induced deposition processes lead to highly contaminated deposition products. Our involvement (WG2 Chair: S. Barth) in an European COST project CELINA resulted in new insigths. The use of heterometallic precursors enables the deposition of an unusually clean metal deposit without subsequent cleaning steps. These significant results sparked additional interest of other groups and extends our network of active collaborations in this area of interest.
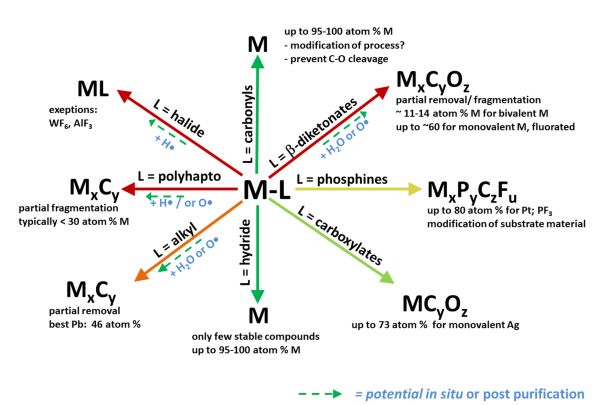
Current understanding of the precursor fragmentation for different ligand classes in precursors being investigated for FEBID can be summarised as follows (and elaborated upon in a review currently under peer review):
 Selected Papers: 1) "Electron Induced Surface Reactions of HFeCo3(CO)12, a Bimetallic Precursor for Focused Electron Beam Induced Deposition (FEBID)" R. K. T P, I. Unlu, S. Barth, O. Ingólfsson, D. H. Fairbrother* J. Phys. Chem. C 2018, 122, 2648–2660. 2) "Electron Interactions with the heteronuclear carbonyl precursor (H2FeRu3(CO)13): from fundamental gas phase and surface science studies to focused electron beam induced deposition " R. K. T P, P. Weirich, L. Hrachowina, M. Hanefeld, R. Bjornsson, H. R. Hrodmarsson, S. Barth, D. H. Fairbrother, M. Huth, O. Ingólfsson* Beilstein J. Nanotechnol. 2018, 9, 555-579. 3)"Magnetic characterization of direct-write free-form building blocks for artificial magnetic 3D lattices” M. K. I. Al Mamoori, L. Keller, J. Pieper, S. Barth, R. Winkler, H. Plank, J. Mueller, M. Huth* Materials 2018, 11, 289. 4) "Formation and decay of negative ion states up to 11 eV above the ionization energy of the nanofabrication precursor HFeCo3(CO)12" F. K. T. R. , R. Bjornsson, S. Barth, O. Ingolfsson* Chem. Sci. 2017, 8, 5949-5952. 5) "Structure and energetics in dissociative electron attachment to HFeCo3(CO)12" K. T. R. Kumar, S. Barth, R. Bjornsson, O. Ingolfsson* Eur. Phys. J. D, 2016, 70, 163. 6) “Direct writing of CoFe alloy nanostructures by focused electron beam induced deposition from a heteronuclear precursor” F. Porrati*, M. Pohlit, J. Müller, S. Barth, F. Biegger, C. Gspan, H. Plank, M. Huth Nanotechnology 2015, 26, 475701. |
Modified Alkoxides for Material Synthesis
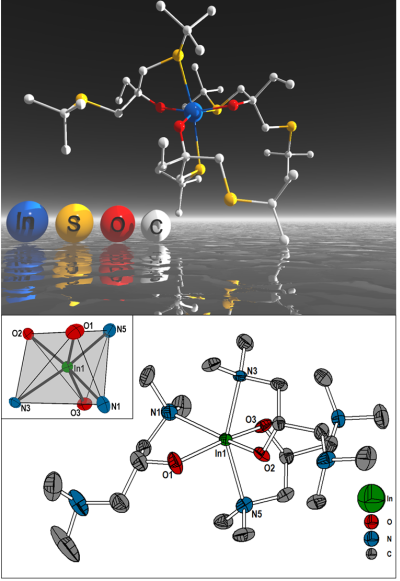
| Metal alkoxides [M(OR)x]y are suitable precursors for oxide synthesis. However, we demonstrated that an additional functionality can be incorporated in the organic backbone of the ligands by introducing thioether functionalities. Steric demand and the possibility of the weakly coordinating thioether functionality allowed us to prepare monomeric group 13 alkoxides and several other derivatives. These compounds allow an in situ conversion of the M-O bonds from the alkoxide in M-S moieties in the thermally derived solids. This enables us to investigate new paths for material synthesis. In addition, aminoalcoholate derivatives have been prepared, which are also monomeric compounds in the solid state.
Selected Papers: 1) "Monomeric aminoalkoxides of aluminium (III), gallium (III) and indium(III)" F. Biegger, S. Barth* Monatsh. Chem. - Chemical Monthly 2016, 147, 361-368. 2) “Synthesis and characterisation of thioether functionalised gallium and indium alkoxides” S. Barth*, F. Biegger, M. Puchberger Dalton Trans. 2015, 44, 16439-16445. 3) “Influence of precursor chemistry on CVD grown TiO2 coatings: differential cell growth and biocompatibility“ J. Altmayer, S. Barth*, S. Mathur* RSC Advances 2013, 3, 11234-11239. |
|
Precursors for the Synthesis of Materials with Metastable Composition
|
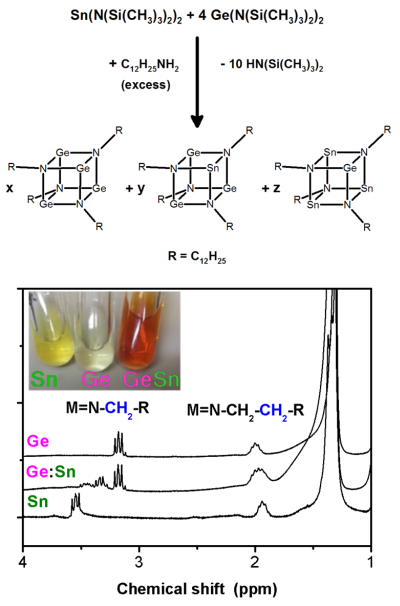
Metal amides and imides are important compounds for the synthesis of metallic nanostructures and metalloids. For instance, the formation of mixed metallic imido cubanes allowed us to synthesize the first bottom-up derived germanium-tin nanowires with a high tin content. The metal silylamides used are converted in the imdo cubane species by ligand exchange. Their formation can be easily followed by NMR spectroscopy and allowed us to get deeper insight in the formation mechanism of Ge nanostructures and Ge/Sn alloys from molecular sources in solution based processes.
Selected Papers: “Microwave-assisted Ge1-xSnx nanowire synthesis: precursor species and growth regimes” M. S. Seifner, F. Biegger, A. Lugstein, J. Bernardi, S. Barth* Chem. Mater. 2015, 27, 6125-6130. |